Pharmaceuticals Recall
Pharmaceutical recalls require fast, precise communication to prevent unsafe products from being consumed. VeriDoc Global can enable instant identification of recalled medicines through secure QR codes linked to blockchain records, ensuring patients and pharmacies always receive accurate recall information in real time.

Explore the complete proof of concept and see the technology come to life.
How Pharmaceutical Recall Verification Works
Reliable Control for Recalled Pharmaceutical Products
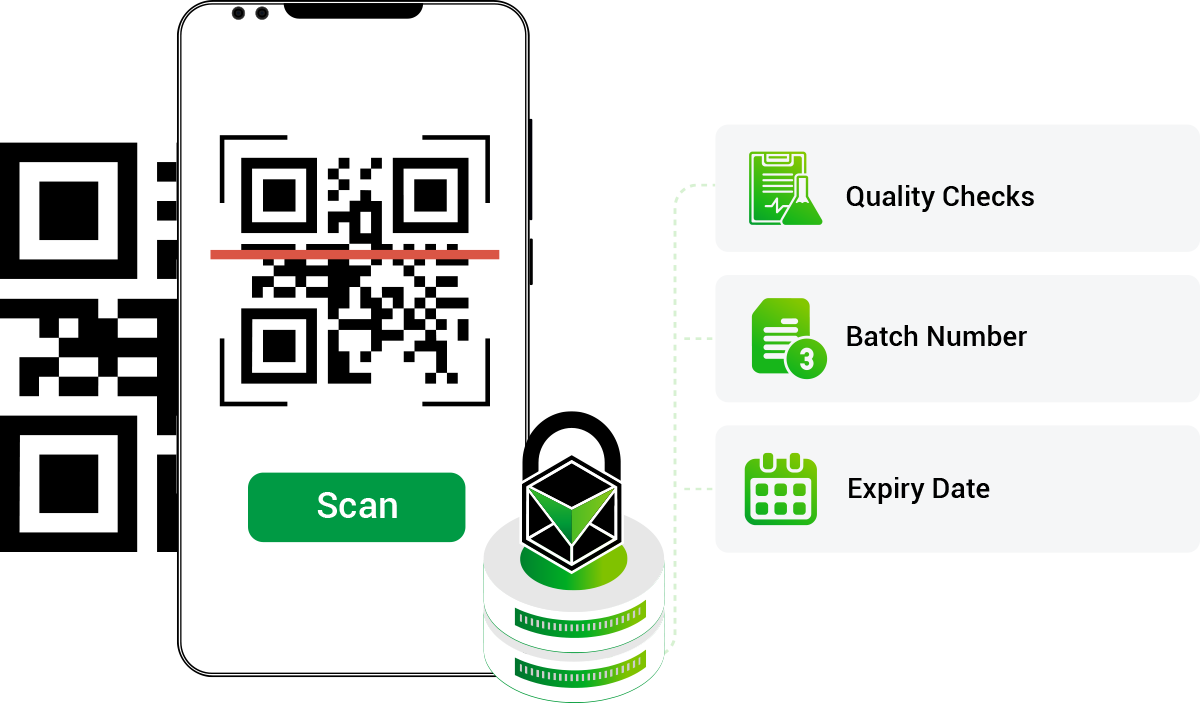
Batch-Level Product Verification
Verification technology allows pharmaceutical companies to confirm the authenticity of specific medicine batches. This helps identify which products are subject to recall and which remain safe for distribution.
Supply Chain Recall Visibility
Distributors and pharmacies can quickly scan product packaging to determine whether a medicine belongs to a recalled batch. This improves transparency and coordination during recall procedures.
Prevent Re-Circulation Medicines
Recalled pharmaceutical products must be removed from circulation to avoid patient risk. VeriDoc Global helps prevent recalled medicines from being redistributed by linking each product package to a unique blockchain verification record.
VeriDoc Global for Pharmaceutical Recall Management
VeriDoc Global provides secure verification technology designed to help pharmaceutical companies maintain product integrity and improve supply chain transparency. In situations such as pharmaceutical product recalls, the platform allows manufacturers to authenticate medicines and confirm whether a product belongs to a specific production batch.
When pharmaceutical products are registered within the VeriDoc Global verification platform, a cryptographic hash is generated from the packaging and product identification data. This digital fingerprint is then stored permanently on the blockchain ledger. Because the blockchain record cannot be altered, the system allows organisations to validate medicines against their original data and identify products that may be affected by a recall.
If counterfeit packaging or modified QR codes attempt to imitate legitimate products, the verification process will detect that the scanned information does not match the original blockchain record. By combining blockchain infrastructure with QR-based verification, VeriDoc Global enables pharmaceutical companies, distributors, and healthcare providers to verify medicines instantly while supporting safer and more efficient recall management.